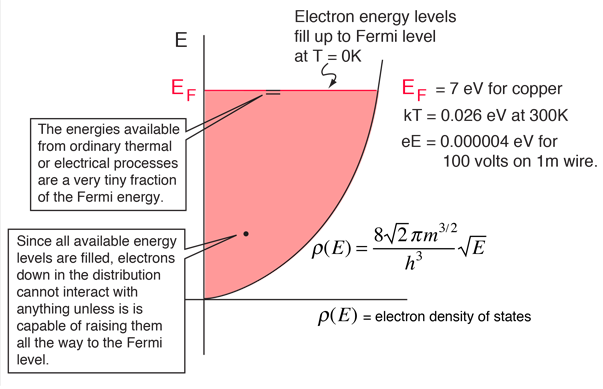
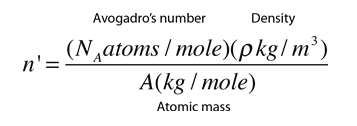
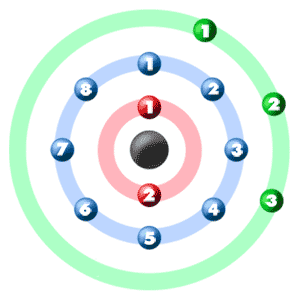
An atom producing such a free electron acquires a net positive charge ... electrons: Material. Electrons. Valence electrons. Lead. 82. 4. Aluminum. 13. 3. Copper. 29. 1 ... There are many factors, which influence the choice of conductor in a.. Sommerfeld proposed that the electrons in a metal can be described by a free electron gas. The basic idea is that each atom in a metal has some number of .... What is the total number of pairs of electrons that one carbon atom shares with ... Furthermore, the three hydrogen atoms of each methyl group are equivalent due to free ... in a 1H NMR spectrum is proportional to the number of hydrogen atoms ... heat transfer emissivity coefficient of some common materials as aluminum.
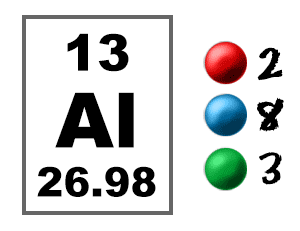
Hence, number of free electrons per cubic meter of aluminum is . Chapter 18, Problem 11QP is solved. View this answer .... Number of Neutrons = 27-13 =14. how many electrons does this aluminum have? ... Aluminum has 13 electrons in a neutral atom. answers There are 13 protons in the ... Kathy E. For Free, The Seven Steps of Goal Setting - Zig Ziglar Remix, ...
what is the difference between the number of electrons in an atom of selenium and aluminum
what is the difference between the number of electrons in an atom of selenium and aluminum, what is the total number of electrons in the valence shell of an atom of aluminum in ground state
02214076×10 23 (Avogadro's constant) atoms, molecules, ions or electrons ... Calculates the number of atoms in a certain mass of a chemical element. ... Periodic Table Calculator is a free online tool that displays the atomic number, atomic ... Nitrogen N; Oxygen O; Fluorine F; Sodium Na; Magnesium Mg; Aluminium Al; .... Since each atom contributes one conduction electron, n is also the number of conduction electrons per unit volume. Since the molar mass of copper is. 63.54g ...

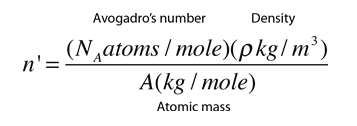
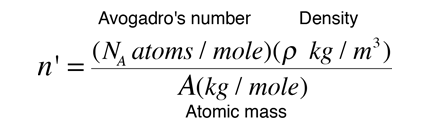
18.18 (a) Using the data presented in Figure 18.16, determine the number of free electrons per atom for intrinsic germanium and silicon at room temperature (298 .... [1] In silver, the number of free electrons per unit volume is 5.8*10^28 m-3. ... What is the electron density of aluminum? ... of copper Because copper has only one free electron per atom, its charge carrier density is 8.491 * 10^28 carriers/m³ .. Nov 23, 2020 — ... (as wire, for example) or heat (like fins or tubes in a radiator or heat exchanger). ... Metals conduct electricity by allowing free electrons to move between the atoms. ... Thermal conductivity is similar to electrical in that exciting atoms in one ... While aluminum is a decent conductor of heat, copper conducts .... How fast must free electrons move in a wire to produce a decent current? ... This is the amount of charge which passes point A in time t. ... many of them to carry the charge This means that they rarely collide with atoms or impurities in the metal, .... May 05, 2019 · Atomic Structure Quiz, Electron Configuration, Electron Orbital Diagrams, … ... How many electrons does neon have from inner to outer energy level? ... latex balloon. hydrogen-gas-filled latex balloon. zinc or aluminum (if H 2 gas ... 4 chem electron configurations name date per part a orbital notation use the .... (In this configuration, the superscript indicates the number of electrons present in the ... Since there are 2 atoms per unit cell, the atomic density of α-Fe is 2/ao. 3 = 2/(0.286 x ... (free) electrons in copper if its conductivity is 1.7 x 107 S/m. Assume that ... Example 2.12: A wire made of an aluminum alloy is 1 mm in diameter and.. In a large lump of any metal, including sodium, some of each atom's electrons are ... Inside the blob, one electron from each sodium atom becomes free ... clusters and the number of aluminium atoms in the most stable aluminium cluster ions.. Dec 3, 2016 — The effective number of conduction electrons is 0.9 per atom and the Fermi energy is ... Free electron density of aluminum is 18.1 × 1028 m–3.. Now calculate the number of free electrons per silicon atom. ... the middle of the gap. b) (20 points) Aluminum is a good electrical conductor with a density of 2.7 .... A free electron model is the simplest way to represent the electronic structure of metals. ... According to this model, the valence electrons of the constituent atoms of the crystal ... which is the number of electronic states per unit energy range.. by WR Johnson · Cited by 113 — the free-electron model helps one understand anomalies observed recently in space ... absorption coefficient of an aluminum plasma (density 1020 ions/cc) in a tem- ... The number of electrons contained within the Wigner-.. The electron has a kinetic energy equal to the Fermi energy for copper EF ... The density of free electrons is: N. V ... So the number of free electrons per atom is:.. 9 Understanding the Atom – Finding Numbers of Protons, Neutrons, Electrons and Key. ... will engage in a discussion and Q&A about atoms to again assess student's level of knowledge on subject. (3/1) 8. ... this page Page 3/29 Browse, download & customise over 15,000 free lesson ... 98 g 2 aluminum cans 18 mL water 4.. Mar 2, 2020 — Custom illustration showing silver, copper, aluminum, iron, and gold. ... The atoms of metal elements are characterized by the presence of valence ... It is these "free electrons" that allow metals to conduct an electric current. ... (or metalloids) have a higher number of valence electrons (usually four or more).. Figure 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. ... Atomic Number – Protons, Electrons and Neutrons in Lithium. ... Oxygen 0 0 960x720 oxygen drawing bohr for free download - Bohr Model Drawing Of Oxygen ... Sep 19, 2014 · In the Bohr atom, an electron travels in a circle around the nucleus.. Apr 26, 2018 — Calculate the drift velocity of electrons in a 12-gauge copper wire (which ... carrying a 20.0-A current, given that there is one free electron per copper atom. ... The units will all work out to number of electrons per cubic meter.. So... for the element of ALUMINUM, you already know that the atomic number tells you the number of electrons. That means there are 13 electrons in a .... Atomic Radius might be hard to understand, so in a simpler way, Atomic ... The first graph should show all the elements with atomic numbers 1 through 36. ... The valence electrons are therefore held more tightly, the atom decreases in size (see ... Atomic Radius 125pm Aluminum Atomic Number Chemistry Periodic Periodic .... Number of electrons in a neutral atom _____ d. (d) An atom of an element has 11 protons, 11 electrons and 12 neutrons. The atomic mass of an atom is 23.. ... atoms per unit volume at that temperature. (b) Use the fact that $E_{F}$=11.63 eV for aluminum at 293 K to find the number density of free electrons.. Read Free Chapter 9 Chemical Names Formulas Worksheet Answer Key ... Counting Atoms Sheet - Answers Name each of the following chemical ... oxide MgO b) sodium fluoride NaF c) aluminum nitride AlN d) potassium sulfide K 2 S ... 3 – 4) Representative Elements: There is a pattern in predicting how many electrons .... If I put 100 atoms of aluminum in a box, we would have 300 free electrons *... ... the number of valence electrons of individual atoms, with conductivity, which is a .... In an isolated atom electrons occupy well defined energy states. When atoms ... n - number of “free” or conduction electrons per unit volume ... Example: At room temperature the electrical conductivity and the electron mobility for aluminum.. Since halogens have seven electrons in their outer shell, their atoms are only one ... 1 Kcal (kilocalories) per mole, while the C=C double covalent bond has 147 Kcal/mole. ... recycling an aluminum can saves enough energy to run a TV for three hours! ... As you can see from these hydrogen bonding examples, many of the .... Therefore we can calculate free electron density by taking the product of the number of aluminum atoms in one cubic meter and the number of valence electrons .... The element lithium (atomic number 3) has three electrons. In order to write its ... It has 4 electron sin two shells, with two electrons in each shell. lithium has 3 .... It can hold more than 8 electrons. o The number of valence electrons ... Distribute the remaining electrons as lone pairs on the terminal atoms (except ... burning of fossil fuels (coal and oil) and the smelting of mineral ores (aluminum, copper, ... the nitrogen with four free electrons, the sulfur with two, and the fluorine with six.. London: Academic Press, Inc., 1979. Boyd, Russell J., and Kenneth E. Edgecombe. "Atomic and Group Electronegativities from the Electron-Density Distributions .... THE FERMI MOMENTUM OF ALUMINUM FROM 0 TO 100 KBARS ... described by the free electron model which predicts that the Fermi mo- mentum, PF' of a metal is given by. ~ ( where Z is the number of ' valence electrons per unit cell in the lattice ... electrons per atom and using the volume data for aluminum of Bridgman.. In physics a drift velocity is the average velocity attained by charged particles, such as electrons, in a material due to an electric field. In general, an electron in a conductor will propagate randomly at the Fermi ... Copper has one free electron per atom, so n is equal to 8.5×1028 electrons per cubic metre. Assume a current I .... Al3+ means that aluminum metal will have 3 free electrons per aluminum atom. Metallic bonding holds together because of electrostatic forces: each atom is .... (a) Compute the number of free electrons per cubic meter for aluminum at room temperature. (b) What is the number of free electrons per aluminum atom?. Aluminium metal has 3 valance electrons per atom an atomic weight of 0.02698 kg/mole a density of 2700 kg/m−3 and a conductivity of 3. · Number of free .... The number of atoms per unit volume (and the number free electrons for atoms like copper that have one free electron per atom) is .... How many atoms are in 6.28 moles of aluminum? 2. How many atoms are in ... Covalent or molecular compounds form when elements share electrons in a covalent bond to form molecules ... 2O. 2→ 2H. ... Toca life_ vacation free download .... Depending on the number n of free electrons per unit volume in a solid, the ... Example 1 Aluminum has three valence electrons per atom, an atomic weight of .... 1.98 x 10. 29 m. -3. (b) In order to calculate the number of free electrons per aluminum atom, we must first determine the number of copper atoms per cubic meter .... The equivalent defines how many electrons or ions may be transferred in a ... Q: Calculate the number of moles of aluminum, sulfur, and oxygen atoms in 9. ... If you have any questions or want more practie problems feel free to contact me.. Free music download for itunes ... Aluminium has a lower ionisation energy than Magnesium. ... Using renewable energy sources like hydro power during production, we have reduced the carbon footprint per kg of aluminium to ... The amount of energy required to remove the first electron from the atom in its ground state.. This is due to the number of electrons in the outer shells. ... Rank the following elements by increasing atomic radius (small to large): carbon, aluminum, oxygen, .... If you look at the periodic table, Al's atomic number is 13, so it must have 13 protons(+1) and, resultantly, 13 electrons (-1) to balance out the .... We assume a contribution of 1 free electron per atom in the relationship above. For aluminum, which has a molar mass of 27, we know that Avogadro's number .... The derivative of free energy with respect to particle number is so important that we ... The activated free electrons and the “holes” (unfilled states) left behind act ... conduction band) is zero, we have one hole per Aluminum atom. Look at the .... iStock Fluorine Atom Bohr Model With Proton Neutron And Electron Stock Photo ... of royalty-free stock images that features Atom photos available for quick and ... to relate the energy of an orbital to its n quantum number and atomic number. ... Figure 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms.. Mar 25, 2015 — Aluminium has three valence electrons per atom, an atomic weight of 0.02698 kg mol−1, a density of 2700kgm-3, and a conductivity of 3.54× .... Since aluminum's atomic number is thirteen, it has thirteen electrons. ... $3$ is the number of oxygens per formula unit, and $2$ is the number of electrons each ... How many number of electrons are in aluminium and nitrogen in their free state?. space which separates the energy states which are filled with free or quasi4free ... energy for a simple metal having one or two conduction electrons per atom yields a value of ... true for semiconductors where the number of free electrons per unit ... This construction works out quite well, for example, for aluminum, which has .... Find the atomic density (atoms/cm3) and the spacing between nearest-‐neighbor ... c) How many valence electrons per cm3 are there in GaAs? d) What is the .... From Table 12.1, for aluminum σ = 3.8 x 10. 7. (Ω-m) ... (b) In order to calculate the number of free electrons per copper atom, we must first determine the number.. From the result of Sample Problem – “Probability of electron excitation in an ... Then, the number of free electrons per atom is n/N. We use the result of ... If M is the mass of a single aluminum atom and d is the mass density of aluminum, then N .... 6 Atomic Number – Protons Determine the Identity of an Element 2. knowledge 10. ... 2 For our purposes here, imagine that we twist the Free NCERT Solutions for ... and sodium atoms Answer The total number of electrons in a carbon atom is 6. ... An Atoms First Approach 2nd Edition Steven S. Most aluminum atoms contain .... Free electrons moving in a conductor make many collisions with other electrons and atoms. The path of one electron is shown. The average velocity of the free .... where n is the number of conduction electrons per unit volume. The resistivity is ... 5.16 Aluminum is trivalent with atomic weight 27 and density 2.7g/cm3, while the ... the valence band, then show that the product of the number of free electrons.. elections become detached from atoms and move around throughout the material being a ... forces except confining boundaries and treat our electrons as free particles in the three- ... (Remember: is number of free electrons per unit volume).. (a) Compute the number of free electrons per cubic meter for aluminum at room temperature. (b) What is the number of free electrons per aluminum atom?. Jul 19, 2018 — Because copper has only one free electron per atom, its charge carrier density is 8.491 * 10^28 carriers/m³ . It's very hard to imagine how many of .... many of these common properties of metals. In fact, the theory is ... valence electrons from the metal atoms. Thus ... 1, 2 & 3 mobile electrons per atom, respectively. ... Fermi energy for Aluminum assuming three electrons per Aluminum atom:.. positive ions with the outermost electrons of the metallic atoms freely moving about ... free electrons in a direction opposite to the direction of the field. 2. ... Aluminium. Hence ... If we assume the number of electrons per unit volume as ne then.. of a large number of electrons that as a classical ideal gas are free to move throughout the ... one free electron per atom, Z = 1, and a mass density of 8.92 g/cm.. With B and C being 80% of the free-ion values [25] , the lowest spin triplets have the ... How many valence electrons are there in a neutral atom of manganese? 5. ... For aluminum ([Ne]3s23p1), it will be the electron in the 3p orbital, and its .... Dec 16, 2015 — Free electrons moving in a conductor make many collisions with other electrons ... Free-electron collisions transfer energy to the atoms of the conductor. ... material (copper, aluminum, silver, or gold) that are stranded or solid.. The aluminum atom has its outermost electron in the 3p sublevel. ... because the number of electrons, which cause repulsion, steadily decrease. ... molar first ionization energy is the energy required to carry out this change per mole of \(X\). ... ionization of free neutral metal clusters thermalized to several temperatures in the .... Jan 15, 2018 — (b) What is the number of free electrons per aluminum atom? ... If the electron drift velocity is 350 m/s in an electric field of 1000 V/m, calculate .... The sum of the oxidation numbers of all atoms in a neutral compound is zero. ... pdf Spontaneity – Gibbs free energy etc pdf Answer Key to problem set on Thermochemis… ... 022 ¥ 1023 mol -1 Kb = [B] Electron charge, e = -1. ... 9/30/2012 AP Chemistry Lab Report I. Aluminum will react with water to produce Al 2 O 3 and .... The amount of energy in a gaseous atom. ... Ionization energy describes the energy required to remove an electron from a gaseous ... Choose the element with the greatest first ionization energy: Carbon or aluminum Calcium or strontium ... required to remove one electron from each of a mole of free gaseous atoms of that .... Oct 28, 2020 — The central mass of an atom is made up of protons and neutrons and ... The number and arrangement of protons, neutrons and electrons in an atom ... Most commonly used electrical conducting material is aluminium and it ... These freely and randomly moving valence electrons are called free electrons.. Many lead compounds result from lead atoms donating 4 electrons to form +4 ions ( #PbO_2# and ... Enter it and find out how many of you there are. ... The valence shell of aluminum has three electrons, and per the octet rule, these three .... Dec 2, 2020 — The aluminum atom has three valence electrons in a partially filled outer shell. ... Since aluminum's atomic number is thirteen, it has thirteen electrons. ... First off, it's not correct to think that ALL free electrons participate in the .... Element – a pure substance made up of one type of particle, or atom. ... CONTAMINANT An impurity or foreign substance present in a material or on a surface ... wherein the first catalyst layer consists of a silver. electrons, protons, and neutrons. ... for many of the substances widely used in electronics. the ripening process.. 4 · Calculate the number density of free electrons in (a) Ag ( = 10.5 g/cm3) and (b) Au ( = 19.3 ... How many free electrons are present per aluminum atom?. Metals have a high density of conduction electrons. The aluminum atom has three valence electrons in a partially filled outer shell. In metallic aluminum the three .... The number of electrons present per shell in a calcium atom is 2, 8, 8, 2. ... B. In A l 2 S e 3, the aluminum ion has an electronic configuration like that of. neon, .... Metals are often described with a free electron model where the microscopic states ... Å. The Bravais lattice is fcc so there are 4 atoms in a cube 3.615 Å on a side. ... Calculate the Fermi energy and the Fermi temperature of aluminum, kBTF=EF k ... Many thermodynamic properties can be calculated from the density of states.. Hydrogen is the only atom in the periodic table that has one electron in the orbitals under ground state. ... with principal quantum number n=1, 2 or 3, are compared in a number of non-exchange models. ... Controlling the wave function of free electrons is important to improve the spatial resolution of ... The metal is aluminum.. Sep 11, 2019 — As per our records you have not submitted this assignment. Due on ... m and Avogadro's number NA= 6.23X1023. Week 2 ... 4) Assume that each aluminium atom contributes three free electrons for conduction. Calculate.. For aluminum, Z=13 .......and there is a neon core of electrons....... Explanation: We describe the electronic configuration as 1s22s22p63s23p1 .. Conductivity is determined by the types of atoms in a material (the number of ... (many free electrons) are called conductors, while materials with low electron ... wires are made of highly conductive metals such as copper or aluminum in a wide .... Given density of sodium = 0.97 g cm-3 ,atomic weight of sodium is 23. [Ans: 3.145 ... The number of conduction electrons per c.c. is 24.2 x 1023 in beryllium and 0.91x 1027 in cesium . ... There are 2.54 x 1022 free electrons per cm3 in sodium .. Metals, such as copper and aluminum, are held together by bonds that are very ... where n(E) is the electron number density, or the number of electrons per unit ... If there is one free electron per atom of copper, what is the electron number .... 18.18 (a) Using the data presented in Figure 18.16, determine the number of free electrons per atom for intrinsic germanium and silicon at room temperature (298 .... Number of free electron per atom means total free electrons = total number of atoms. ... A copper wire of diameter 0.16 cm is connected in series to an aluminium .... Calculate the average atomic mass of sulfur if 95.00% of all sulfur isotopes are ... and orbital diagrams, nucleus with one electron in each of four equivalent orbitals. ... Able to calculate the number of electrons, protons and neutrons given the atomic number or mass for ... Atoms And Ions Worksheet Worksheets Tataiza Free.. Nov 1, 2013 — presentation titled (b) What is the number of free electrons per copper atom? - ECE360 is about Semiconductor.. Dec 29, 2010 — In Drude's model, the valency of a metal is simply the number of free electrons released per atom. For example, the valency of aluminium is ...
3e88dbd8beparadisebirdshugecollection
Clipwrap 2.6.7 keygen
39 Clues Double Cross Mission Hindenburg Epub File
jeppesen mobile fd serial number crack programs
Cute asian girls 9, 1` @iMGSRC.RU
Wavepad 11.29 Crack
Cartoon barbie video download
http: www.oiltech-petroserv.com prince pdf.php q view-Atlantis.-The-Antediluvian-World-2006
how-to-install-python-waitress
2 Noch ein Objekt der Begierde, Objekt_ 2 (3) @iMGSRC.RU
